I need the effective density of states near the valence band maximum/conduction band minimum for the n-type, p-type, and intrinsic Si wafer. Could you offer me this information?
Learn The Difference Between Conduction Band and Valance Band?
What Are The Density of States Near The Valence Band?
A PhD candidate requested a quote for the following:
Question:
Answer:
The effective density of states near the valence band maximum (for p-type) and conduction band minimum (for n-type) in silicon (Si) wafers are important parameters in semiconductor physics. For an intrinsic (undoped) Si wafer, both n-type and p-type properties are relevant. The values depend on the effective mass of the charge carriers (electrons and holes) and the temperature.
The effective density of states in the conduction band (N_C) and valence band (N_V) for silicon at room temperature (around 300 K) are typically given as:
- N_C (conduction band, relevant for n-type Si): Approximately 2.8 x 10^19 cm^-3
- N_V (valence band, relevant for p-type Si): Approximately 1.04 x 10^19 cm^-3
For an intrinsic Si wafer, both N_C and N_V are relevant, and the intrinsic carrier concentration (n_i) can be calculated using these values along with the bandgap energy of silicon and the Boltzmann constant.
Please note that these values are approximate and can vary slightly based on the specific conditions, such as temperature and doping levels. For precise calculations or for conditions significantly different from room temperature, it's recommended to use more specific data or formulas that take into account these variations.
Reference #250233 for more or click here!
Get Your Quote FAST! Or, Buy Online and Start Researching Today!
Which N-Doped Wafer Has Highest Electric Conductivity?
The electrical conductivity of an n-doped wafer depends on the material used and the level of doping. Generally, silicon and gallium arsenide are common materials for wafers. In terms of the highest electric conductivity, heavily n-doped silicon or gallium arsenide wafers are often considered among the best. The doping element, such as phosphorus or arsenic for silicon, significantly influences the conductivity.
However, the "highest" electric conductivity can vary based on the specific application and the exact doping levels and methods used. Advanced materials like graphene or silicon carbide might also exhibit high conductivity in certain conditions. For the most accurate information, it would be advisable to consult specific data sheets or studies related to the material and doping levels of interest.
Arsenic (As) and antimony (Sb) doped silicon wafers are used in different applications based on their specific electrical and physical properties:
-
Arsenic-Doped Silicon Wafers:
- High-Speed Electronics and Microprocessors: Arsenic is commonly used in high-speed electronics and microprocessor applications due to its ability to create highly conductive n-type regions. Its high solubility and fast diffusion properties make it suitable for forming shallow, heavily-doped junctions that are essential in modern high-performance and high-density integrated circuits.
- Power Devices: Arsenic doping is also used in power devices, where high conductivity and efficiency are crucial. These applications benefit from arsenic's ability to create low-resistance contacts and regions.
- Antimony-Doped Silicon Wafers:
- High-Temperature and High-Power Applications: Antimony is preferred in applications that require stable operation at high temperatures or in high-power environments. Due to its lower diffusion rate compared to arsenic, antimony maintains a more stable dopant profile at elevated temperatures, which is important in power devices and applications where thermal stability is crucial.
- Precision Devices and Sensors: In applications where precise control over dopant profiles and concentrations is required, such as certain types of sensors and precision devices, antimony's lower diffusion rate makes it a suitable choice. This allows for more accurate control over the electrical characteristics of the device.
- High-Temperature and High-Power Applications: Antimony is preferred in applications that require stable operation at high temperatures or in high-power environments. Due to its lower diffusion rate compared to arsenic, antimony maintains a more stable dopant profile at elevated temperatures, which is important in power devices and applications where thermal stability is crucial.
Both dopants are used in the semiconductor industry to tailor the electrical properties of silicon wafers for specific applications. The choice between arsenic and antimony depends on factors such as the required electrical characteristics, thermal budget, and fabrication processes involved in the particular application.
Which N-Type Silicon Has the Higest Electrical Conductivity?
-
Highest Electrical Conductivity of Semiconductors: It is not universally accepted that Sb-doped silicon has the highest electrical conductivity among all semiconductors. Electrical conductivity in semiconductors is influenced by various factors, including the type of dopant, doping concentration, and the semiconductor material itself. Different materials and doping levels can lead to varying conductivity levels.
-
Band Gap and Donor Level: Silicon's band gap is indeed around 1.12 eV at room temperature, but the significance of the Sb donor level being 0.039 eV below the conduction band isn't directly related to it being "ideal for transistors." Transistor performance depends on various factors, including carrier mobility, recombination rates, and more.
-
Dual Plasma Method and Sb-Sn Alloy Nanocrystals: This part of the statement seems unrelated to the electrical conductivity of Sb-doped silicon. The method for synthesizing Sb-Sn alloy nanocrystals is a specific research topic and does not directly pertain to the general properties of Sb-doped silicon wafers.
-
Effect of Natural Oxide and SOD Doping: The presence of natural oxide on silicon and its impact on electrical characteristics is a complex topic. While doping can affect conductivity, the specific conductivity values and the effects of SOD doping mentioned are not standard and may vary based on experimental conditions.
-
Four-Point Probe Measurement: This technique is indeed used to measure electrical conductivity, but the specific behavior described (increase at 0.228 J of laser energy, etc.) is highly specific and not a general characteristic of Sb-doped silicon.
-
Dopant-Site Bonding Energies (EB): The concept of dopant-site bonding energies is valid, but the specific values and comparisons (e.g., boron in silicon) are context-dependent and not necessarily indicative of the overall performance of Sb-doped silicon.problem of surface segregation of resistive phases.
Reference #270670 for specs and pricing.
What is the Difference Between Conduction Band and Valance Band?
The conduction band and valence band are key concepts in the field of solid-state physics, particularly in the study of semiconductors and insulators. Here's the difference between them:
-
Valence Band:
- The valence band is the highest range of electron energies in a solid where electrons are normally present at absolute zero temperature.
- It consists of electrons that are bound to atoms and participate in the formation of chemical bonds.
- These electrons are responsible for the electrical properties of the material in its ground state.
- When electrons are in the valence band, the material behaves as an insulator or a
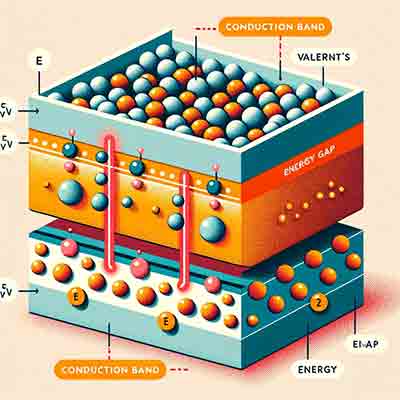 semiconductor, depending on the energy gap between the valence band and the conduction band.
semiconductor, depending on the energy gap between the valence band and the conduction band.
-
Conduction Band:
- The conduction band is the range of electron energies higher than the valence band, where free electrons can exist.
- These electrons are not bound to atoms and can move freely through the material, contributing to electrical conductivity.
- In conductors, the conduction band overlaps with the valence band, allowing electrons to move freely even at low energies. In semiconductors and insulators, the conduction band is separated from the valence band by an energy gap.
- When electrons gain enough energy (e.g., through thermal excitation or photon absorption) to jump from the valence band to the conduction band, they contribute to electrical conduction.
The key difference lies in their role in electrical conductivity: electrons in the valence band are involved in bonding and do not contribute significantly to conductivity under normal conditions, whereas electrons in the conduction band are free to move and contribute to the electrical conductivity of the material. The energy gap between these two bands determines the electrical nature of the material (conductor, semiconductor, or insulator).
